
Anatomy & Physiology
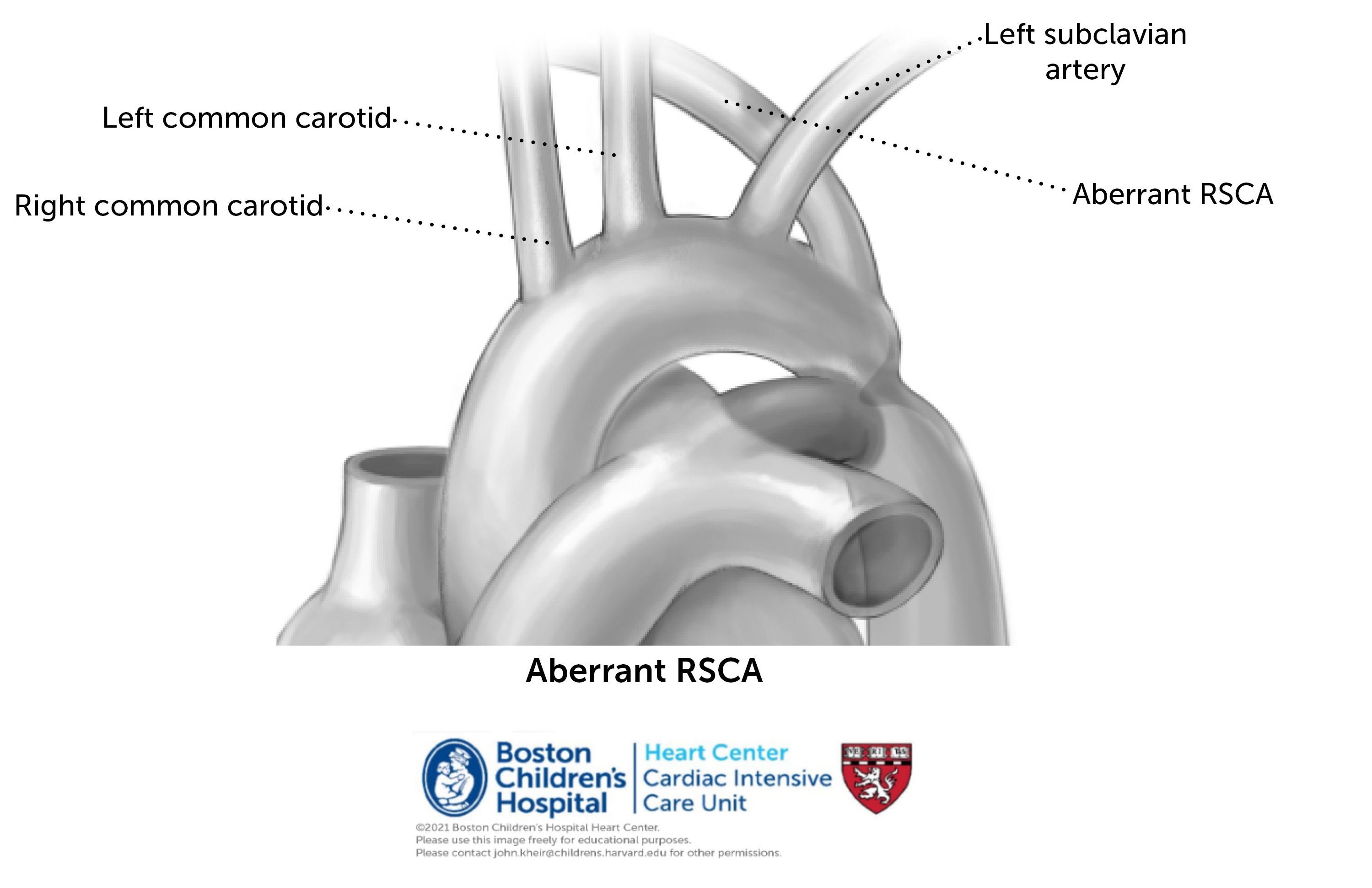
Patients with coarctation of the aorta and no other associated cardiac anomalies are said to have an ‘isolated CoA’ (coarctation of the aorta). Like every heart disease, CoA is a spectrum that can range from mild to critical; a critical coarctation is purely defined as one that is dependent on the presence of a PDA for flow to the lower half of the body to be patent. The presence of a PDA permits a pathway for blood to pass ‘around’ the coarctation, which is really a involution of scar tissue at the aortic isthmus. In the presence of a VSD, blood flow into the aorta is disfavored (higher resistance of the aortic circulation with a ventricular level shunt –> more left to right flow in the ventricles), predisposing to more diffuse arch hypoplasia, or even an interrupted aortic arch (IAA). Alternative arch anatomies include a ‘bovine’ arch and aberrant right subclavian artery (seen below).

Spectrum of possible preoperative clinical courses
- Prenatal diagnosis and immediate PGE: Patients are generally stable after birth so long as the PDA remains open and widely patent. Ensuring the presence of distal pulses and a 4 extremity BP gradient <10 mmHg (means) is important. Ensure patient is on PGE through a functional IV. Generally, oral feeding is acceptable if the patient is clinically well.
- Post-natal diagnosis and early PGE: CoA is sometimes identified postnatally, often through a failed pulse oximetry screening (one that demonstrates post-ductal desaturation). In this case, the PDA is usually open and often unrestrictive, such that there is no ischemic insult from lack of blood flow to the lower half of the body. Clinical course should be approximately the same as above, although some patients may have had renal/mesenteric ischemia due to decreased flow, such that labs and careful attention to be paid to identify these. Some patients may also have a degree of LV dysfunction, which can be identified on echo and treated with inotropes.
- Post-natal diagnosis with presentation of shock state: In other cases, a neonate or infant presents in shock, often at the time of ductal closure (few days to weeks of life). Symptoms may include increased work of breathing, vomiting, poor feeding, hypoglycemia, coagulopathy, absent pulses, or even cardiac arrest. CXR may show pulmonary congestion due to left atrial HTN. This presentation may be dire, as there is often a significant burden of ischemic injury to the liver, bowel and kidneys, severe metabolic acidosis, and ventricular dysfunction (presumably due to high afterload and acidosis). Treatment should include immediate PGE initiation. At times a higher dose than normal is used to more rapidly open a closed PDA (normal starting dose is 0.01 micrograms/kg/minute, may increase 5-10 fold). Depending on the presentation, patients may also require include intubation and mechanical ventilation (to alleviate work of breathing, decrease VO2 and redistribute lung water), sedation and neuromuscular blockade, early echocardiogram (important to confirm the diagnosis of critical CoA and rule-out other anomalies that can present similarly), inotropic support, and diuretics (to decrease lung water). Monitor renal function and for clinical signs of NEC. Enteral feeding of any kind may increase mesenteric oxygen demand and should be done with caution once acidosis has resolved and exam is reassuring. It is vital to ensure that PGE is infusing correctly through a well-functioning IV at the prescribed dose.
- Late diagnosis: In some cases, a coarctation is not critical, meaning the patient has survived for time (at times years) with a closed PDA. Generally, these patients have extensive collateralization around the site of coarctation, although there is typically a significant blood pressure gradient between upper and lower extremities that leads to diagnosis. Ventricular function may or may not be normal, and ventricular hypertrophy may also be present. Patients typically present to the ICU postoperatively only and are in generally good health preoperatively.
Operative interventions
Simple end-to-end anastomosis (ETEA)
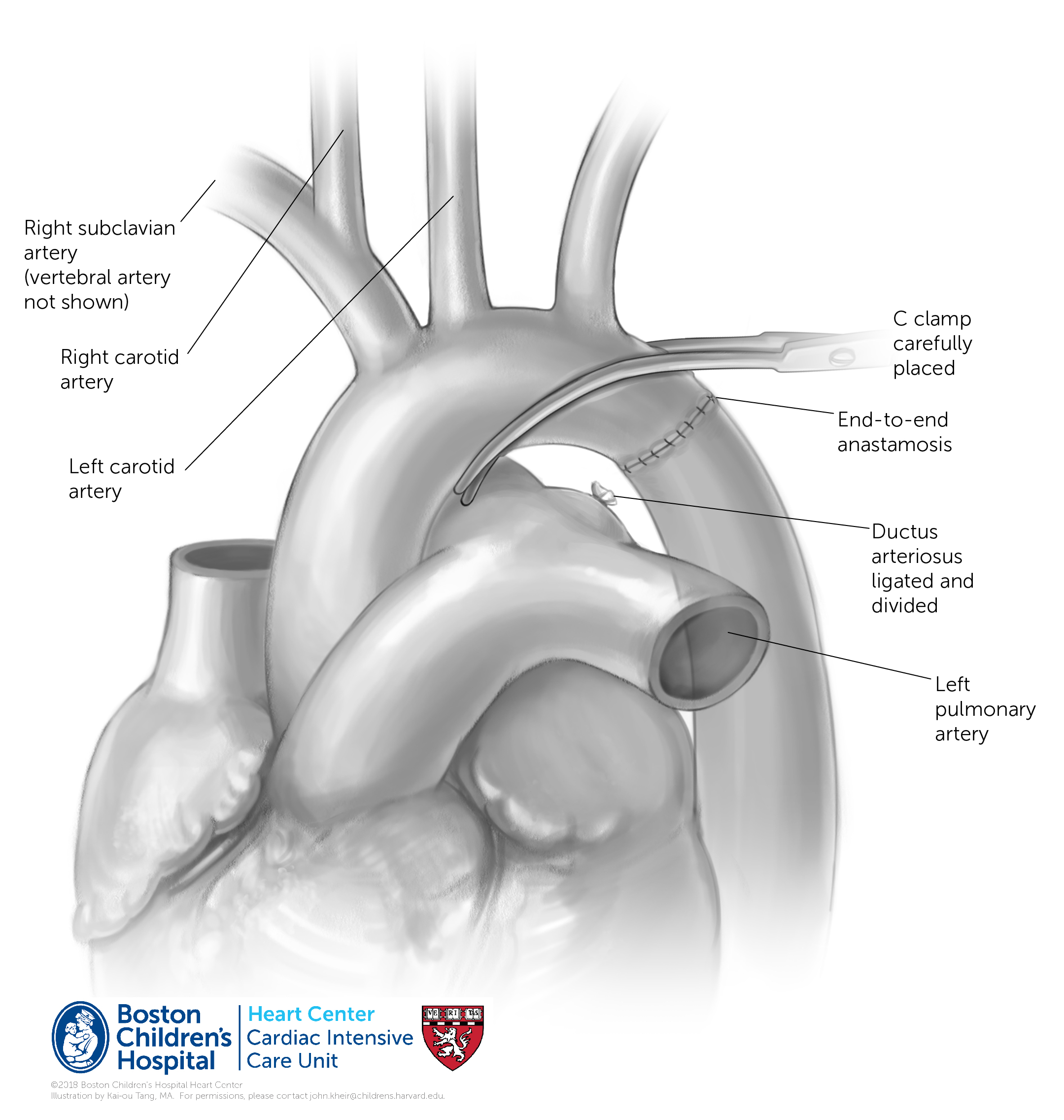
If there are no complicating comorbidities or anatomic considerations (e.g. RAA), a coarctation resection (i.e. coarctectomy) is often performed without cardiopulmonary bypass via lateral thoracotomy. The transverse aorta is cross-clamped with care to ensure antegrade blood flow into all of the brachiocephalic arteries. The coarctation is resected entirely with the ductus, which is ligated and divided on the pulmonary end. At times the left subclavian artery is ligated and divided to help mobilize the aorta to decrease tension on the suture line. Rarely, the left subclavian can be used as a flap to augment the aortic isthmus. Generally, the aorta is cross-clamped for 15-30 minutes for the procedure, which takes place at ~33-35°C (ambient cooling only). Blood flow to the brain is antegrade, to the spinal cord is through vertebral and intercostal arteries. Postoperative gradient is assessed by echo, and often by comparison of a right radial arterial line with lower extremity cuff BP. Four extremity BPs should be performed postoperatively on a regular basis particularly in patients who are not doing as expected. Rarely, residual ductal tissue may cause a recurrent coarctation. Injury to the thoracic duct, recurrent laryngeal nerve (vocal cord function), and vagus nerve are possible and should be monitored for.
Extended end-to-end anastomosis (extended ETEA)
Often, the transverse aortic arch is sufficiently hypoplastic that it would cause a gradient following a traditional ETEA. Such cases may be associated with a VSD (as shown), in which case they would necessarily be repaired using CPB. In the absence of a VSD, a CoA with hypoplastic transverse arch may be repaired via thoracotomy (‘from the side’) with clamp placement as shown. This procedure differs from a simple ETEA in that the underside of the aortic arch is incised and augmented using the oblique incision of the descending aorta.

Postoperative course spectrum
- When preoperative status is favorable, extubation generally takes place within 12 hours. Monitor for chylothorax and vocal cord dysfunction, which may manifest as aspiration with feeding. These two comorbidities may significantly complicate the postoperative course.
- When a significant degree of obstruction was present preoperatively (particularly in older patients), refeeding should take place slowly and with close monitoring for mesenteric angina. A refeeding syndrome with feeding intolerance is not uncommon in this situation, and can be severe in its presentation (e.g. acute abdomen).
References
2015, JTCVS – Aortic arch variants [PDF]
2008, JTCVS – arch anatomy and wall stress [PDF]
2006, Am J Neuroradio – Arch anatomies [PDF]
1989, Circulation – Long-term followup after CoA repair [PDF]
1987, Circulation – CoA/VSD importance of malalignment [PDF]